Background
Electrocatalysts play a key role in fuel cells, water electrolyzers, and other devices related to sustainable energy conversion. However, the most efficient catalysts today contain noble metals such as platinum or iridium. To enhance the technoeconomic viability of hydrogen-related technology, we must develop efficient catalysts requiring less noble metals.
Currently, the dominant method to identify promising materials is Density Functional Theory (DFT). This simulation technique yields information on the binding energies between the catalyst and key intermediates. Based on this information, the thermodynamic efficiency of the candidate material can be evaluated. Recent works incorporating machine learning have succeeded in identifying promising materials from millions of candidates.
However, the main criteria of materials evaluation is thermodynamics, which contains no insight on time scale information. The most important catalytic properties, such as catalytic rates or material lifetime, are fundamentally questions about time scale. This is why we believe that understanding the kinetics of catalytic reactions is important, not only to identify promising candidates, but also to truly understand the behavior of catalysts under real operating conditions. Our research group will pursue this goal via the following three directions.
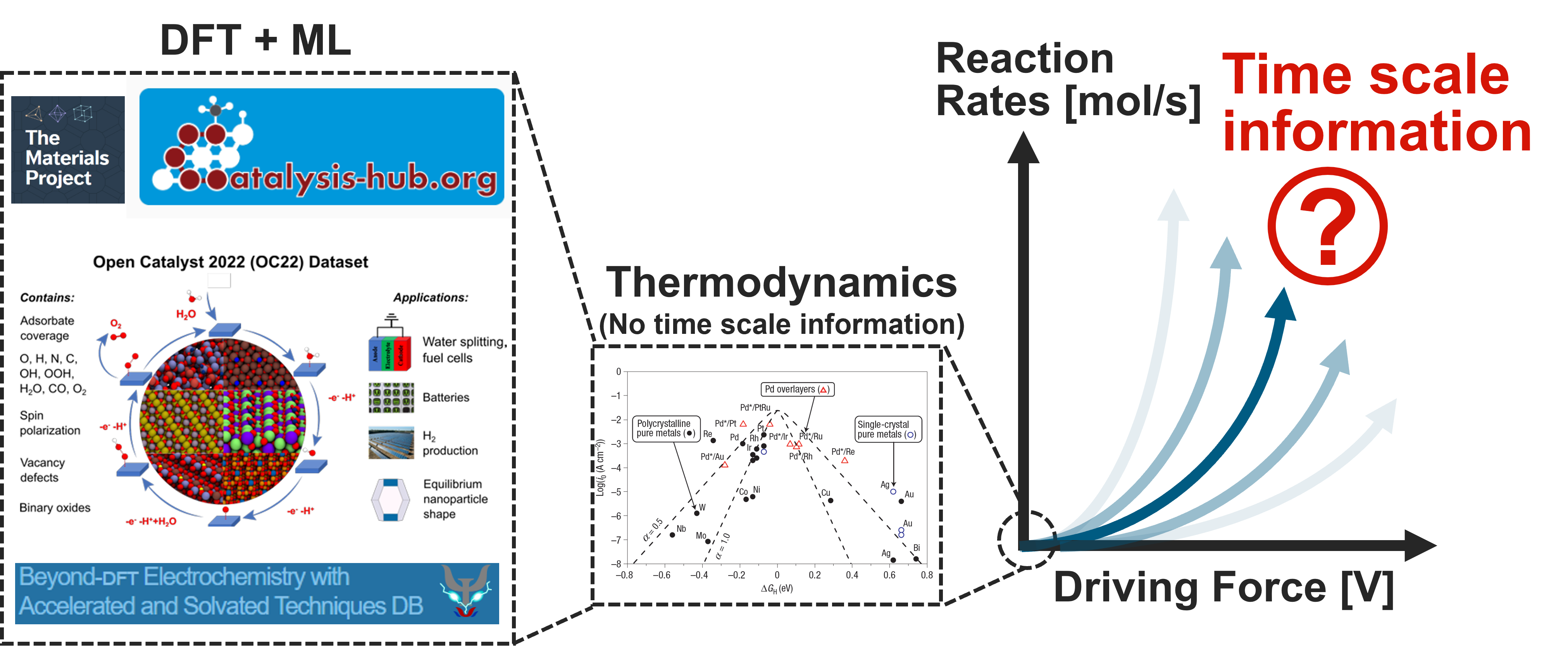
Comparison between the dominant, DFT-based approach and our approach based on kinetics.
Research Directions
Overview
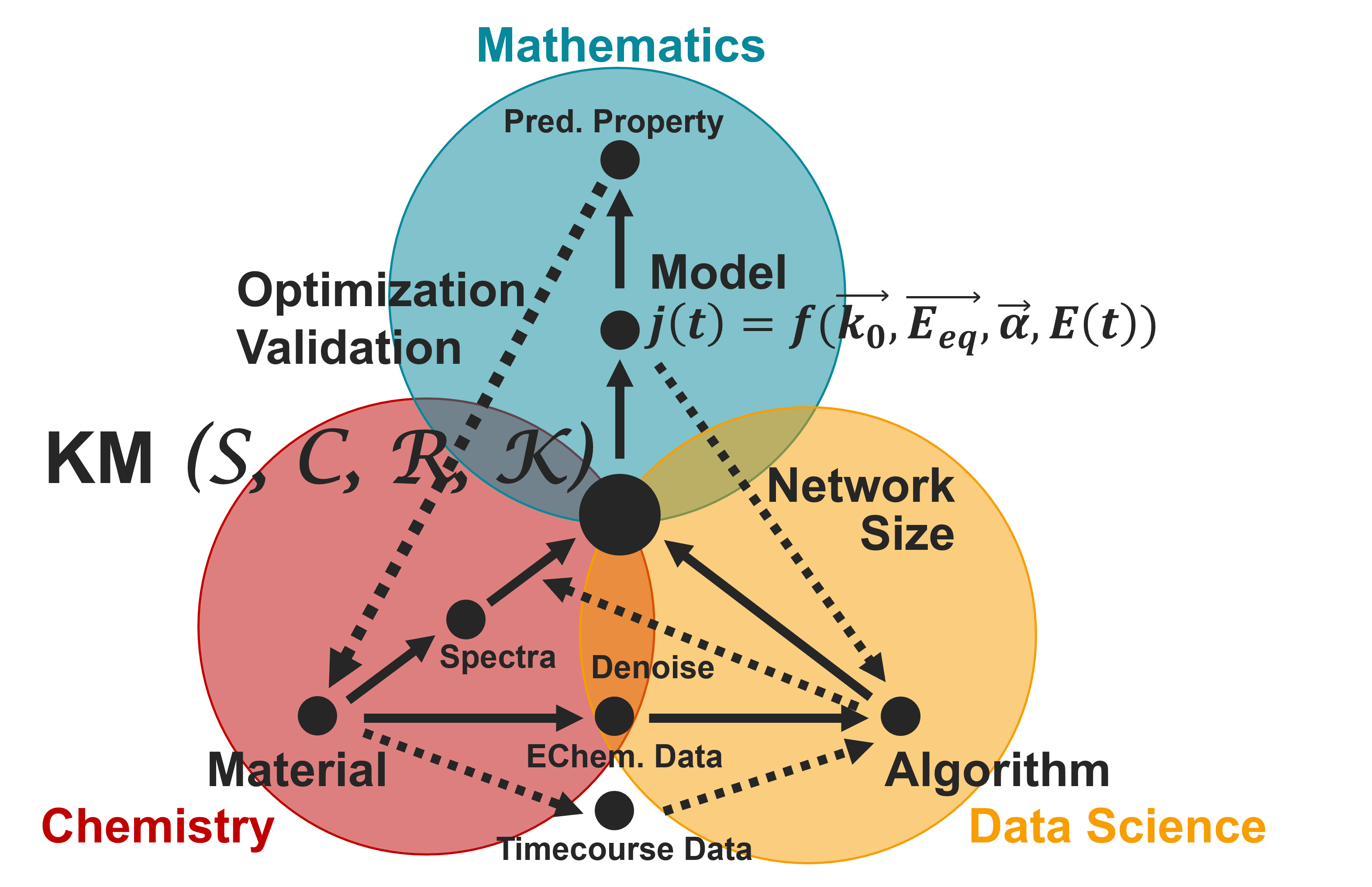
An overview of the work in this group, which bridges applied mathematics, electrochemistry, and data science. KM: Kinetic Mechanism.
- Developing Models to Predict Catalytic Properties
- Developing Advanced Materials Using Microkinetics
- Developing Algorithms to Identify Rate Constants
In electrocatalysis, the most important catalytic properties are summarized in the current density j(t) and how it changes with time. A large current indicates an active catalyst. The timescale at which the current can be maintained is the operational lifetime of the material. The selectivity towards side reactions can be evaluated by comparing partial current densities towards the main product and the side product (for example, OER vs CER).
These properties ultimately depend on the kinetic mechanism, which is a combination of the intermediate species, the elementary reactions, and their rate constants. Therefore, in this project, we will derive new mathematical formulae which allows catalytic properties (activity, stability, selectivity) to be calculated from the kinetic mechanism. All of our work so far has been focused on mass-action kinetics. However, we are also incorporating more physical models to understand electrocatalytic deactivation via mechanisms such as dissolution and delamination.
Selected Publications: ChemSusChem 2025, JPCL 2024, JPCL 2019.
In this project, we will develop top-notch materials for electrocatalytic reactions, thus allowing us to make direct contributions to sustainability. By doing so, we will demonstrate the importance of kinetics in catalysis.
So far, much of the previous literature has focused on the catalytic performance at low current densities (mA/cm2 order). Under these near-equilibrium conditions, material properties resemble the thermodynamic information obtained from DFT. However, real applications require current densities larger by 2 or 3 orders of magnitude (A/cm2 order). Under these conditions, kinetic factors such as Tafel slopes (how much a reaction is accelerated by applying additional voltage) cannot be ignored. We believe that the gap between screening conditions and actual applications has lead to many promising materials to be overlooked.
Additionally, we are currently trying to speed up materials evaluation using kinetic simulations. Extending material lifetime using a purely experimental approach is not easy, because each test may take weeks or months. Furtheremore, better materials require longer tests because they are more stable. To speed up this process, we are developing algorithms to identify the kinetic mechanism from minute-scale measurements (see next project). If we could use the kinetic insight to predict catalytic lifetime, this would allow us to direct our experimental efforts to only the most promising materials, which would greatly promote materials development.
Selected Publications: JPCL 2024.
One of the main reason most works in catalysis don't focus on kinetics is because information on rate constants are difficult to obtain. Activation barrier calculations from DFT have errors of at least 0.03 eV. This converts into a 5 fold error in rate constants, which translates directly to errors in reaction rates and material lifetimes. No kinetic predictions can be made without accurate rate constants; thus the lack of rate constants is a big challenge towards understanding the kinetics of catalytic reactions.
To address this challenge, we are developing new data-analysis techniques to obtain rate constants from experimental data. Our previous work used the genetic algorithm to fit HER Tafel plots using microkinetic equations. Recently, we are developing methods to fit OER Tafel plots, as well as non-steady-state electrochemical measurements, time-course data, and in-situ spectroscopy data. In addition to obtaining rate constants, these algorithms are also important to verify the validity of the proposed mechasmim. Namely, for a mechanism to be kinetically plausible, there must be at least one set of rate constants which can reproduce the experimental dataset.
Selected Publications: ACS Catalysis 2021.
Core Expertise
- Kinetic Analysis
- Optical Waveguide (OWG) Spectroscopy
Our lab specializes in developing data-analysis algorithms to identify the mechanism of chemical reactions. We also have experience in handling large amounts of spectral data such as UV-Vis and small-angle X-ray diffraction (SAXS). If you have large volumes of data and are unsure of what to do with them, perhaps we can help. We also have ongoing work on denoising and fitting spectra obtained from electrocatalytic reactions.
Selected Publications: Nat. Commun. 2024, JPCL 2024, ACS Catalysis 2021.
The second core expertise of our lab is optical waveguide (OWG) spectroscopy. Conceptually, it is like an in-situ ATR UV-Vis: It is a surface sensitive technique, and the depth profile of a sample can be obtained by changing the angle of the incident light. It can obtain spectra at the millisecond order, making it perfect to determine the mechanism of electrocatalytic reactions. If you are interested in measuring your sample, please let us know.
Perspective and Other Directions
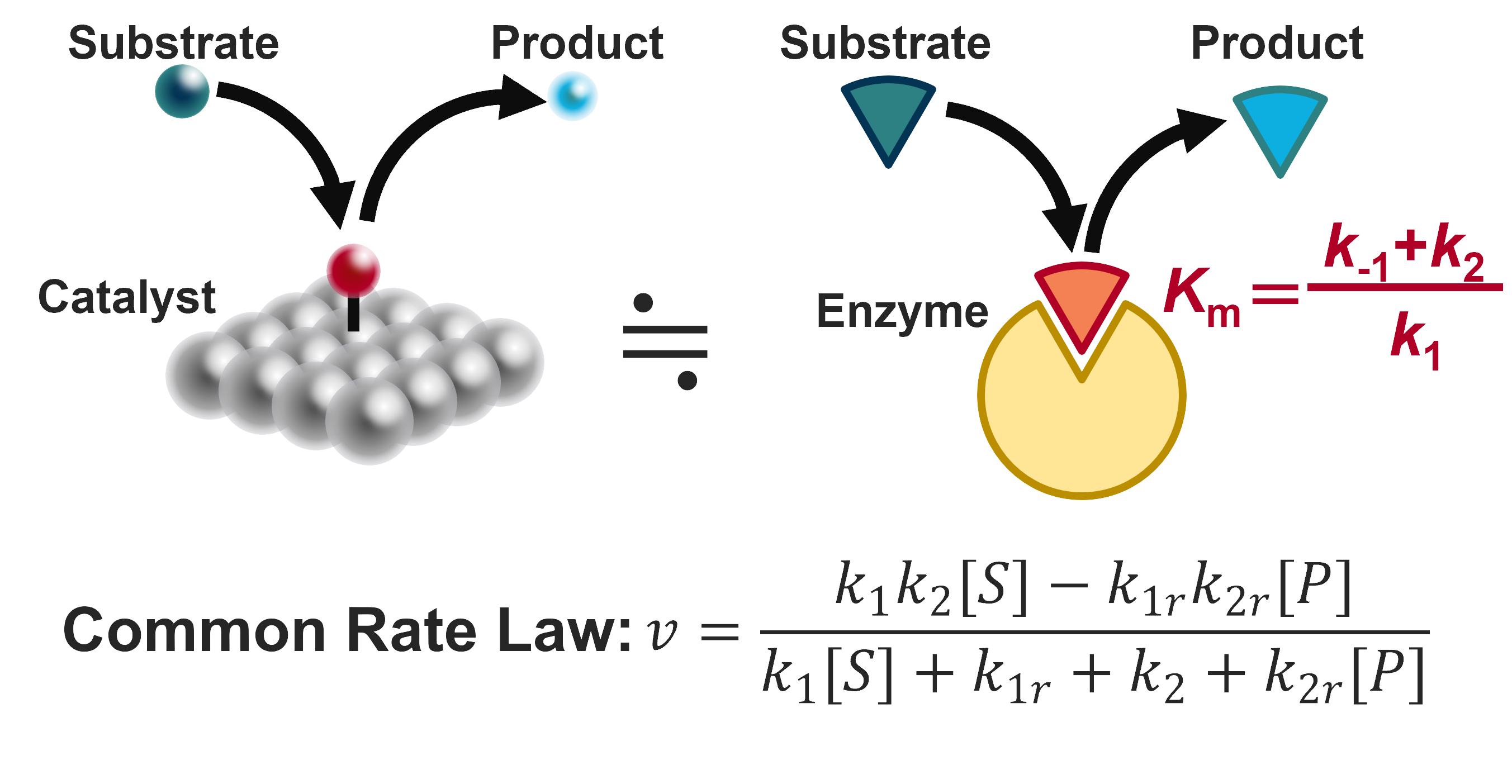
Similarity between catalysts and enzymes.
The directions listed above are all targeted towards improving electrocatalysts. However, the approach of focusing on the kinetic mechanism and then using them to make predictions is not restricted to electrocatalysts. In particular, Michaelis-Menten type enzymes have only a single intermediate and therefore, their reaction mechanism and corresponding rate equation is almost identical to the hydrogen evolution reaction. Based on this insight, we have previously proposed that the Michaelis-Menten constant Km, which is a measure of the binding affinity, should be equal to the substrate concentration for maximium activity. My bioinformatic analysis on a dataset of 1000 wild-type enzymes have Km values close to the substrate concentration, suggesting that even nature seems to respect the principle Km = [S] (2023 Nat. Commun.). We are actively collaborating with experimentalists, and are looking forward to further exciting collaborations, both inside and outside the immediate field of electrocatalysis.
Selected Publications: Nat. Commun. 2023, Angewandte 2024, Biosci. Biotechnol. Biochem. 2025.